Theranos Trial Reveals Details of Federal Investigation at Troubled CA COVID Lab, Newsom Admin. Renews No-Bid Contract
SACRAMENTO (CBS13) — California's controversial $1.7 billion COVID testing lab contract with PerkinElmer was allowed to auto-renew on Sunday, despite the fact that the lab hasn't fulfilled the terms of its initial contract and is the subject of ongoing state and federal investigations. Those investigations were prompted, in part, by a CBS13 whistleblower investigation.
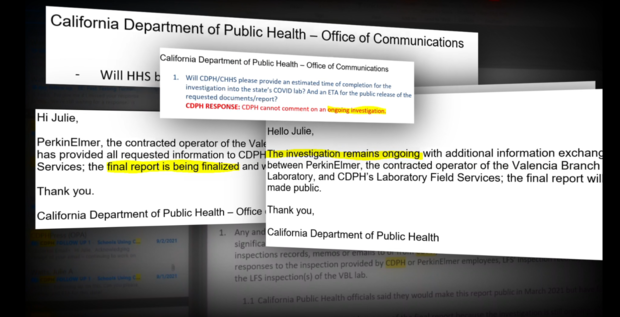
Regulators have refused to release reports of their investigations into flawed tests at California's COVID lab ahead of its contract renewal. However, in an interesting twist, we're learning more about what inspectors found at the state lab, thanks to the unrelated trial of Theranos founder Elizabeth Holmes.
Court Records Indicate:
- Federal inspectors found "serious deficiencies" at California's COVID lab.
- Deficiencies included inaccurate test results, quality control failures, and inadequate validation reports.
- CDPH-PerkinElmer COVID Lab Director's license is in jeopardy.
The Connection Between Theranos & the COVID Lab
The state's lab director, Dr. Adam Rosendorff, is the former lab director of Holmes' disgraced finger-prick blood-testing company, Theranos.
Rosendorff was a key whistleblower in the Theranos case and is a key government witness against Holmes—his former boss at Theranos.
However, the whistleblower is now the subject of a separate whistleblower investigation in his current role as lab director at the California Department of Public Health's (CDPH) state-funded PerkinElmer COVID testing lab.
Court records reveal that Dr. Rosendorff's license is in jeopardy, not because of the flawed tests he oversaw at Theranos, but because of what investigators found at the CDPH-PerkinElmer COVID lab.
PerkinElmer hired Rosendorff in January 2021 to run the state lab. It went live three months earlier and the two original CDPH lab co-directors resigned shortly after the troubled launch.
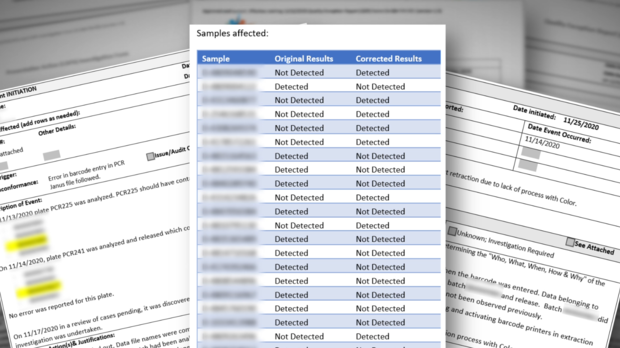
In February, whistleblowers revealed personnel problems at the state's lab ranging from unlicensed and untrained lab techs watching videos and sleeping while processing COVID-19 samples, to test swabs found in lab restrooms.
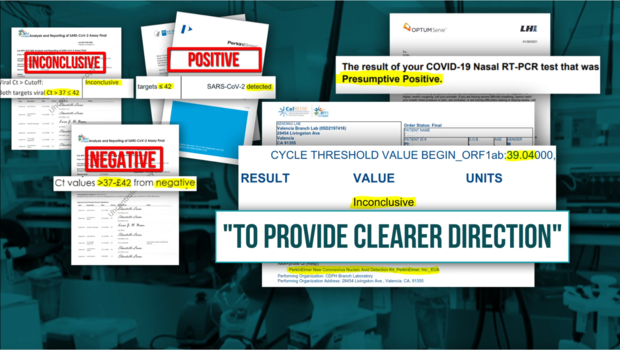
As more whistleblowers came forward, concerns multiplied to include incidents of contamination, swapped samples, wrong results, and tens of thousands of invalid or inconclusive COVID-19 tests.
Whistleblowers also raised concerns about the reliability of the state's PerkinElmer COVID test itself and the validity of the test results.
As we reported, the lab management deviated from the FDA Emergency Use Authorized (EUA) test that is stipulated in the state contract and, instead, began experimenting with its own "Lab Developed Test" (LDT).
CDPH repeatedly denied that the lab was no longer using the EUA test-- until the FDA confirmed to CBS13 that the lab's testing procedures were not authorized under their EUA. In response, CDPH said it had been mistaken and acknowledged it had transitioned to a lab-developed test.
But as we reported, lab-developed tests must go through a lengthy validation process before they can be used on patients. Whistleblowers alleged the test being used on patients at the state lab was not properly validated.
According to court records in the Holmes case, it appears federal regulators may have had similar concerns.
Theranos Trial Confirms Continuing Problems at CA COVID Lab
In an attempt to discredit Rosendorff's testimony about flawed tests at Theranos, Holmes' defense attorneys brought up the federal investigation into faulty tests at his current lab.
Holmes' attorneys told the court that federal lab inspectors from the Centers for Medicaid and Medicare Services (CMS) found "serious deficiencies within the (state's COVID-19) lab," including "inaccurate test results," "quality control failures" and "inadequate validation reports."
They added that the deficiencies had not been corrected as of spring and summer of 2021.
Similarly, CDPH state inspectors reported finding "significant deficiencies" at the lab back in December of 2020.
The state publicly acknowledged those deficiencies in February 2021, following our initial reports, and PerkinElmer claimed at the time that "the deficiencies had "long since been resolved".
However, Holmes' attorneys said, according to CMS, deficiencies continued.
As of October 5th, 2021 Holmes' attorneys said they believed Rosendorff was still at risk of losing his license due to an "immediate jeopardy notice and his inability to cure those deficiencies."
Rosendorff testified on October 5th that he understood he was at risk of having his license suspended "for at least two years."
Conflict of Interest Concerns Between Federal Inspectors and State Lab Director
The court also acknowledged a potential conflict of interests, or bias, in the Holmes case between the federal inspectors who are investigating the state lab and the state's lab director.
The CMS inspectors, to whom Rosendorff provided information about Theranos, and who are testifying alongside him on behalf of the government, are the same CMS inspectors who inspected Rosendorff's own work at the CDPH-PerkinElmer lab and who may now determine if he keeps his license.
Incidentally, CMS has not responded to our Freedom of Information Act requests or emailed questions in over five months.
Whistleblowers have also raised conflict-of-interest concerns related to the state's CDPH investigators who are investigating their own state lab. They've repeatedly asked for an independent audit of the lab.
CDPH, Health Secretary Ghaly Ignore Request to Delay Lab Contract Renewal
"We will continue to press until we get the answer that the public deserves," Senate Republican leader Scott Wilk said in response to the CDPH contract renewal.
He added that he's spoken with Democratic lawmakers who are also concerned.
Wilk had asked the Newsom Administration to delay renewing the no-bid contract with PerkinElmer until the state and federal investigations into ongoing problems at the lab are complete.
"To have a $1.7 billion contract be automatically renewed when we don't even know what happened with the first contract, that's just irresponsible behavior with taxpayers' money," Wilk said.
In the meantime, the state's own public data reveals that the lab hasn't fulfilled the promises made when Gov. Gavin Newsom announced PerkinElmer's no-bid contract.
In his August 2020 press conference, Newsom assured taxpayers,
"What is significant in this partnership is we are demanding test results back within 24 hours, the latest 48 hours, and we have provisions in the contract to guarantee that turnaround time."
But consistent 24-hour test results never came to fruition.
In fact, more than a year later, in the last two weeks of September 2021, the lab returned between one-third and two-thirds of COVID results in more than 48 hours and was among the slowest COVID labs in the state.
While the state lab, known as the Valencia Branch Laboratory (VBL), improved its turnaround time in the few weeks leading up to the contract renewal, the lab still took nearly twice as long to return results as the state average for much of October.
The Unknown Cost to Taxpayers
Taxpayers spent more than $100 million to build out the lab itself and are contracted to pay facility and overhead costs on the lab building in addition to the $1.7 billion-dollar contract for PerkinElmer to staff and run the lab.
CBS 13 has repeatedly requested records related to the total spent on the lab so far, above and beyond the PerkinElmer contract, however, the state has not provided them. It is clear that the $1.7 billion-dollar contract represents a fraction of the total cost to taxpayers.
Though PerkinElmer hasn't yet been paid in full because they haven't fulfilled the contract.
At its peak, the lab processed roughly a quarter of the promised capacity of 150,000 tests a day, and state data reveals that as of August, roughly 1 out of every 42 tests at the lab did not return a clear result.
According to the state, PerkinElmer has been paid $716 million to date to operate the laboratory, "with the majority of funds recovered through the federal funds and health insurance claims."
We've repeatedly asked CDPH and CHHS for records indicating how much money the state has actually recouped from health insurers and federal funds. The state said it did have records responsive to our request but has not provided them.
Other companies now charge a fraction of PerkinElmer's contracted rate with faster results and the state's contract includes provisions that allow the state to cancel its contract if COVID testing becomes "commercially available at lower cost" and Perkin can't match the price.
Still, the state chose to renew its existing no-bid contract anyway.
No Response
The California Department of Public Health did not respond to our detailed questions about the contract renewal but did provide a written statement ahead of the renewal:
"For additional context, the state developed a hub-and-spoke model where the laboratory is connected to over 4,700 specimen collection sites. This includes schools, churches, clinics, and other community sites that are doing the specimen collection and utilizing the laboratory for specimen processing. In fact, over 1,600 of the specimen collection sites are schools conducting PCR testing through the laboratory with another 1,700 schools that are using the laboratory for reflex testing: this further supports our ability to ensure that our children are back in the classroom."
However, as parents have pointed out, faulty and delayed tests from the state lab have kept healthy students out of the classroom, in some cases, unnecessarily quarantined for weeks.
Additionally, the state had a "hub-and-spoke" model prior to building the lab. Until November 2020, CDPH used existing private labs to process the tests from collection sites across the state.
At the time, early in the pandemic, the Governor said the state was paying $100-200 dollars per test, which he said would drop to $30.78 per test on average under the contract with PerkinElmer.
Now, a year and a half into the pandemic, private labs are charging a fraction of the cost of Perkin's contracted rate and are processing far more tests in half the time.
CDPH added:
"We opened the laboratory at a time when testing was limited to laboratories and was a scarce resource both within California and across the nation. Our goal was to get wider and deeper into the community to ensure testing was accessible to the most vulnerable. Of the roughly 2,300 specimen collection sites performing only PCR testing, roughly 68 percent of tests performed at the laboratory are among racial minorities with 32 percent in the most disadvantaged neighborhoods based on the California Health Places Index (HPI) Quartile 1 and 25 percent in Quartile 2."
This, again, raises concerns that these minority and disadvantaged groups are reliant on tests from a lab that, according to court testimony, had been unable to correct serious deficiencies more than six months after they were initially found and remains under investigation by state and federal regulators nearly a year later.
CBS13 specifically asked CDPH if they, CHHS, Secretary Ghaly, the Governor, PerkinElmer, or Dr. Rosendorff would like to comment on Dr. Rosendorff's testimony during the Theranos-Elizabeth Holmes trial. We did not receive a response.
Neither Senator Pan nor Assemblymember Wood—Chairs of the Senate and Assembly health committees—responded to our request for comment on the contract renewal. The Governor credited both men with being instrumental in the lab's development and both voiced their support during the Governor's August 2020 press conference.
As Newsom said when he positioned the PerkinElmer deal as one that would revolutionize COVID testing:
"There's an old saw that says, 'if you continue to do what you've done, you'll get what you've got.'"
A year later, it appears California taxpayers will continue to get what they've got.