CBS News Live
CBS News Sacramento: Local News, Weather & More
Watch CBS News



It's a sight not seen in Sacramento skies for more than twenty years. For the second week in a row, an entire air wing of large military jets landed at McClellan Park.

Loved ones of Kirill Trush want justice for him after he was stabbed and killed near Tower Bridge in West Sacramento over the weekend.

A new rule will affect frozen breaded and stuffed raw chicken products that appear to be fully cooked but are only heat-treated.

The Indianapolis Colts took the first defensive player in the NFL draft, choosing edge rusher Laiatu Latu of UCLA with the 15th pick.

The historical society in Wheatland is making a push to preserve a little-known wagon trail that marked the end of the Donner Party.

Gavin Newsom was asked to comment about Harvey Weinstein's 2020 rape conviction being overturned on Thursday, and the California governor didn't mince words.

A DUI suspect arrested after a Lathrop crash late Thursday night blew a blood-alcohol level of more than three times the legal limit, police say.
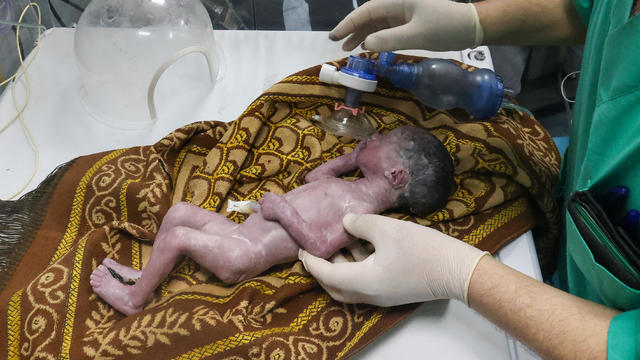
Sabreen Erooh had survived an emergency cesarean section after her mother was fatally wounded in an Israeli airstrike.

The National Highway Traffic Safety Administration is investigating whether last year's recall of Tesla's Autopilot driving system did enough to make sure drivers pay attention to the road.

The Sacramento Canines' Rat Annihilation Team is a group of volunteers that hunt for rats on city streets.

Tens of thousands of students have passed through Victory Elementary School in Stockton since 1923. One former student from the Class of 1942 was there.
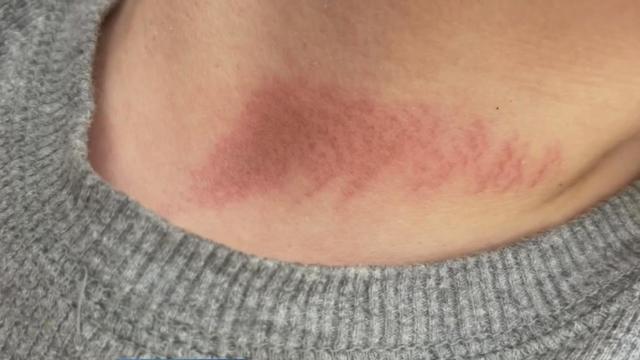
A mother says she has now pulled her child out of a Fairfield school after he was allegedly choked with a jump rope by another student during an after-school program.

Watts will raise awareness of serious issues impacting Californians, hold local officials accountable, obtain answers for viewers and provide solutions.

California law says genetic testing companies have to get your permission before they store, use or sell your DNA, but the state itself doesn't have to get your permission and has been storing DNA samples from every baby born there since the '80s. Lawmakers want to change that, but face an uphill battle.

A bill introduced in the wake of our "Handcuffs in Hallways" investigation aims to reduce "unnecessary" calls for police at schools. But one California lawmaker could kill it without a vote.

Gavin Newsom was asked to comment about Harvey Weinstein's 2020 rape conviction being overturned on Thursday, and the California governor didn't mince words.

This week, things reached a boiling point as demonstrations turned violent over calls for a ceasefire and for campuses to cut ties with companies supporting Israel.

Sacramento, and much of California, received unfavorable grades in this year's State of the Air report from the American Lung Association.
A new rule in California aims to limit health care price increases to just 3% each year.

A former Gridley teacher pled no contest to having sex with an 8th-grade student on the day of his graduation in 2021, according to the Butte County District Attorney's Office.

Find out what kind of weather we're expecting this weekend.

The hunts are done with purpose: to leave the community better than they found it.

No injuries were reported.

One student says the university has not fixed emergency phones or blue lights at her apartment complex.

It's a sight not seen in Sacramento skies for more than twenty years. For the second week in a row, an entire air wing of large military jets landed at McClellan Park.

San Francisco took Florida wide receiver Ricky Pearsall with the 31st pick in the NFL draft on Thursday night to give the 49ers possible insurance if they can't sign Brandon Aiyuk to a long-term deal.

The Oakland Athletics beat the New York Yankees 3-1 Thursday night for a four-game split.

The Indianapolis Colts took the first defensive player in the NFL draft, choosing edge rusher Laiatu Latu of UCLA with the 15th pick.

In 1961, Ed Dwight was selected by President John F. Kennedy to enter an Air Force training program known as the path to NASA's Astronaut Corps. But he ultimately never made it to space.
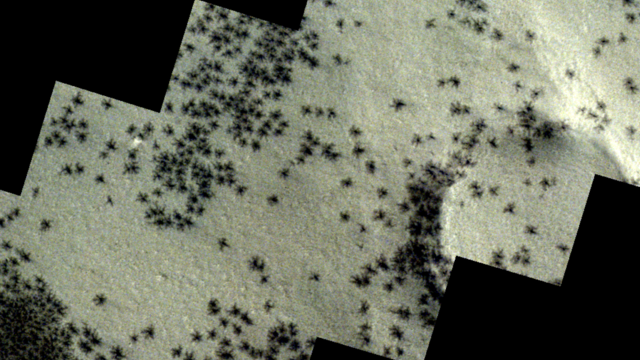
The creepy patterns were observed by the European Space Agency's ExoMars Trace Gas Orbiter.

In November 2023, NASA's Voyager 1 spacecraft stopped sending "readable science and engineering data."

Gavin Newsom was asked to comment about Harvey Weinstein's 2020 rape conviction being overturned on Thursday, and the California governor didn't mince words.

Coal-fired power plants would have to capture smokestack emissions or shut down under a new EPA rule the industry says would make the grid less reliable. It's likely to face court challenges.

The Supreme Court convened to consider whether former President Donald Trump is entitled to broad immunity from criminal charges in the 2020 election case.

A new rule in California aims to limit health care price increases to just 3% each year.

Governor Gavin Newsom is seeking a bold new step to open California medical facilities to Arizona doctors for abortions on their patients after the state's Supreme Court upheld an 1864 abortion ban.

The victims are smart, they're savvy, and they're still getting tricked.

The state regulates crematoriums for people, but we've learned there is zero oversight for pet crematoriums.

Kurtis Ming has had conversations with people in our community about the impact of inflation on their personal finances. It's forcing some to make some tough choices.

A new rule will affect frozen breaded and stuffed raw chicken products that appear to be fully cooked but are only heat-treated.

Sabreen Erooh had survived an emergency cesarean section after her mother was fatally wounded in an Israeli airstrike.

The Indianapolis Colts took the first defensive player in the NFL draft, choosing edge rusher Laiatu Latu of UCLA with the 15th pick.

The Las Vegas Raiders drafted Georgia's Brock Bowers with the 13th pick of the first round on Thursday, the second year in a row they have taken a tight end high in the NFL draft.

In 1961, Ed Dwight was selected by President John F. Kennedy to enter an Air Force training program known as the path to NASA's Astronaut Corps. But he ultimately never made it to space.

Organizers of the annual Outside Lands Music Festival at San Francisco's Golden Gate Park revealed the lineup for the 2024 event.

Mary J. Blige, Cher, Foreigner, A Tribe Called Quest, Kool & The Gang, Ozzy Osbourne, Dave Matthews Band and Peter Frampton have been named to the Rock & Roll Hall of Fame.

Taylor Swift broke her own records, Spotify said, and now owns the record for the top three most-streamed albums in a single day.

The singer was found deceased at her home, a representative said.

Anticipation was growing at a fever pitch before Taylor Swift's latest album, "The Tortured Poets Department," dropped at midnight EDT. But it turned out it's actually a double album.

A new rule in California aims to limit health care price increases to just 3% each year.

Governor Gavin Newsom is seeking a bold new step to open California medical facilities to Arizona doctors for abortions on their patients after the state's Supreme Court upheld an 1864 abortion ban.

Federal officials say they're double checking whether pasteurization has eradicated the danger from possible bird virus particles in milk.
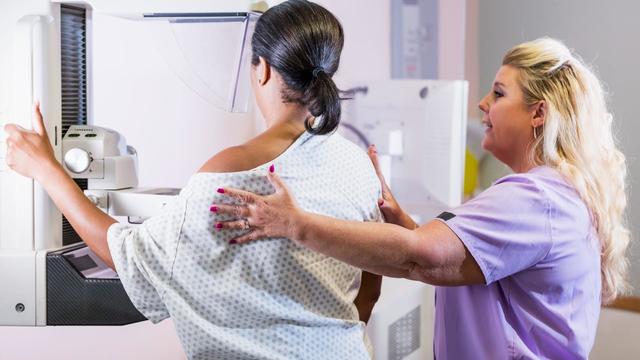
A new CDC study found that not enough women are up to date with their mammograms.

UnitedHealth said it paid the criminals behind attack that crippled hospitals and pharmacies to protect sensitive patient data.

Amazon announced Monday that it suspended its drone delivery operations in the California Central Valley community of Lockeford.

The victims are smart, they're savvy, and they're still getting tricked.

Google began removing California news sites from some user's search results, a test that acted as a threat should the state pass a law requiring the search giant to pay media companies for linking to their content.

Long lunch lines, friendly hellos, and a greater social atmosphere – could all of that return to downtown Sacramento with the return of state workers? Some businesses seem to think so.

A California lawmaker introduced a bill that would end exclusive control of ticket sales by companies like Ticketmaster.

Amazon announced Monday that it suspended its drone delivery operations in the California Central Valley community of Lockeford.

Google confirmed it has fired additional employees for being involved in protests at the tech company's Sunnyvale and New York City offices to oppose a $1.2 billion contract with Israel.

The victims are smart, they're savvy, and they're still getting tricked.

California lawmakers on Monday rejected a proposal aimed at cracking down on how some of the nation's largest utilities spend customers' money.

A bill in California wants to make more rental housing available to tenants with pets.

Two animal welfare measures are moving forward at the California State Capitol with the goal of humanely controlling evergrowing pet populations.

A mountain lion snatched and killed a cat overnight in Calaveras County, the pet's owner told CBS Sacramento on Thursday.

A Natomas woman said the typical trip to the dog park ended in tragedy, with her pet being attacked and killed.

A suspect is sought after 73 dogs were seized across three properties in the city of Turlock last week, authorities said Tuesday.

A Sacramento family is sharing their story after an unexpected delivery in the middle of the night. Someone left a malnourished puppy at their front door and ran off.
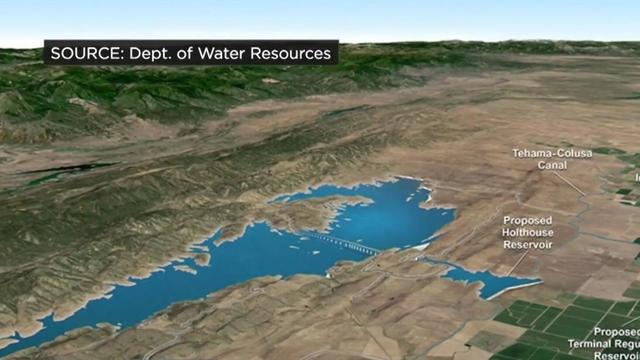
The conversation surrounding California's water continues. The Sites Reservoir project northwest of Sacramento has a price tag of $4 billion and is funded by local, state and federal dollars.

The Regional Water Authority is working on creating a "bank" to store water underground doubling the capacity of groundwater stored.

There's one Northern California reservoir that hasn't rebounded despite this winter's rain and snow.

If you want to measure the success of this winter, California's largest reservoir is a good way to do it. As of Friday, Shasta is about three feet away from full, and that's a foot higher than where it was on Monday.

The DWR upped its water release amount this week. On Wednesday 18,000 cfs were released and 20,000 cfs on Thursday.

A Yuba City mom started a baseball team for kids with physical and intellectual disabilities after finding her 11-year-old son with Down Syndrome didn't have anywhere to play.

A Sacramento nonprofit is teaming up with a local credit union to give back so seniors can safely do basic tasks like bathing or exercising. Those lending a helping hand are also a group of seniors.

The high school players have trained hard all season to make Friday night's finals, and they didn't let their physical and mental challenges get in the way of playing a good game.

NFL Hall of Famer Tim Brown is hosting his 23rd youth football camp at Sacramento State, an annual event that is part of a mentorship program for underserved young people.

An inspiring love story is still being written chapter by chapter at the Brookdale Folsom home for seniors. That's where Faye Guerrero and Rob Trueax found love again after loss.